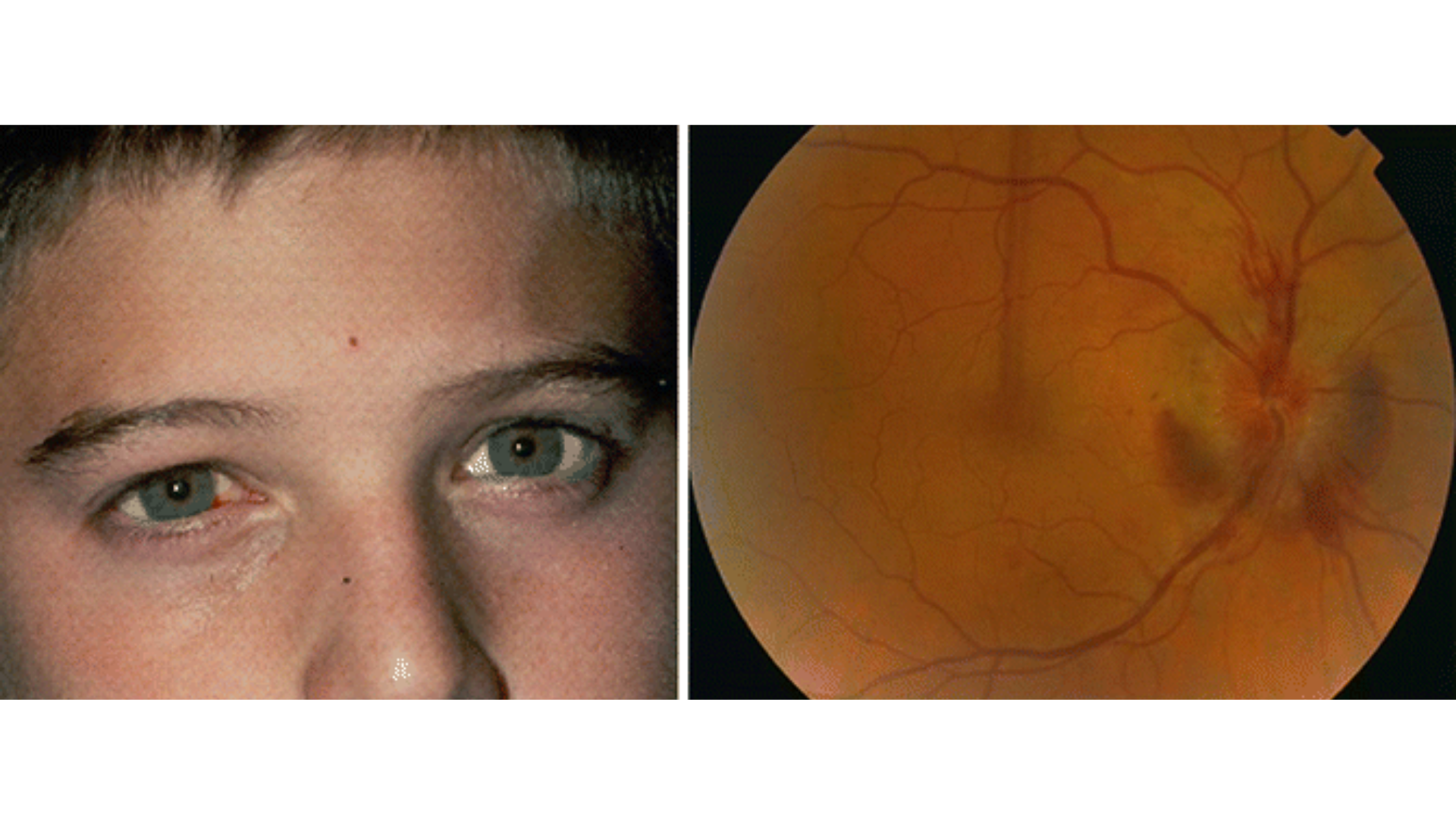
Traumatic optic neuropathy is one of the most devastating causes of permanent visual loss and complete blindness. After a traumatic injury, such as the optic nerve crush, prominent signs of Retinal Ganglion Cells (RGCs) apoptosis are found in the injured retinas. Oxidative stress is considered a pathologic factor that mediates the post-trauma process of RGCs apoptosis. A pioneering study found that hydrogen can promote the survival of RGCs and enhance visual function recovery in an optic nerve crush. Moreover, the terminal deoxynucleotidyl transferase dUTP nick end labeling staining and malondialdehyde assay suggested that the neuroprotective effects were mediated by suppressing the ROS-induced apoptosis.
Hydrogen can exert antioxidant and anti-apoptotic activities via selectively neutralizing the cytotoxic ROS, such as the ·OH and ONOO–. Meanwhile, the signaling ROS playing metabolic roles, such as the superoxide and hydrogen, are far less affected. Unlike some antioxidant supplements with strong reductive reactivity, hydrogen neither disturbs the physiological oxidation reactions nor disrupts the essential defensive mechanisms.

Hydrogen can penetrate the membranes and diffuse into organelles (eg, the mitochondria and nuclear DNA), and thereby get access to the intracellular source of cytotoxic ROS. Based on the favorable distribution characteristic, hydrogen is highly effective for reducing the level of cytotoxic ROS, which impair the nuclear DNA and mitochondria. From the aspect of safety, hydrogen is advantageous to many other antioxidants as it shows no 0cytotoxicity even at high concentrations. These advantages indicate that the application of hydrogen therapy might act as a safe and effective strategy against ROS-related disorders without giving rise to serious side effects.
Call Now: 1800-102-0908
Website: https://www.kykindia.com
Source: https://www.ncbi.nlm.nih.gov/pubmed/24915536







 WhatsApp us
WhatsApp us